Scientists Discover Breakthrough Method To Convert Plastic Waste Into Acetic Acid Using Sunlight …
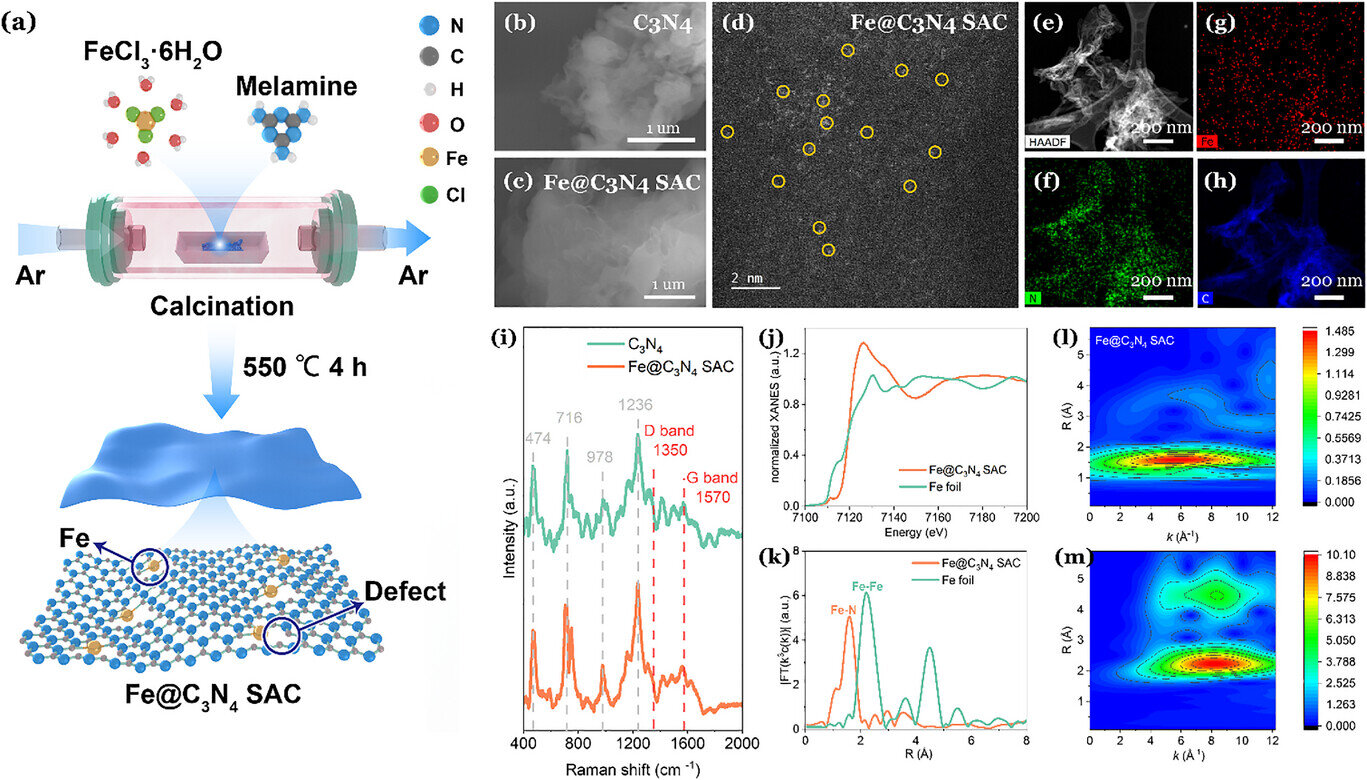
Plastic floats on the surface of our oceans. It chokes the tides. But the sun provides a solution today. Dr. Yimin Wu at the University of Waterloo has found a way to transform this debris into acetic acid. I noticed that his team used iron atoms to mimic the enzymes found in forest fungi. The sun hits the water and the chemical bonds snap.
It is a quiet victory. I think the transition from a discarded bottle to a kitchen staple marks a shift in how we view the lifecycle of our belongings.
The science happens in liquid. It requires no smoke stacks. Sunlight hits a material constructed from carbon nitride. Iron atoms inside the structure trigger a cascade of reactions.
This mimics biological digestion. But the catalyst does not tire. It breaks the long carbon chains of polymers. What resonates with me most is the simplicity of the components. Water. Light. Iron. Scraps of PVC. These findings appeared this month in the journal Advanced Energy Materials. This hits home for me because it suggests the mess in our environment is not a permanent scar.
The laboratory used mixed piles of trash.
They fed the reactor polyethylene. They added polypropylene. They threw in PET containers. The mixture did not slow the reaction. Acetic acid emerged as the primary result. This chemical helps manufacture food. It creates dyes. It powers industries. According to phys.org, the process avoids the fumes usually associated with burning plastic.
I noticed that the efficiency remains high even with messy, real-world waste. And that matters for the health of our rivers. It seems to me that the team has found a way to bypass the expensive sorting that usually stops recycling in its tracks.
The sun is a pair of scissors. It cuts through the stubborn bonds of plastic.
It leaves behind a useful liquid. We are watching chemistry catch up to the efficiency of the natural world. This is a story of human ingenuity meeting the light of day. The reaction works. There is a path forward for the plastic we once thought was immortal.
Plastic floats. I noticed that sunlight can do more than bleach a bottle on the sand.
The reaction occurs in water at room temperature. The beauty of this is that the sun provides every ounce of energy required to snap the carbon bonds. But the method does not stop at clean plastic. It handles the filth of a gutter. I noticed that the team fed the reactor polyethylene. They added polypropylene. They included PET scraps.
The catalyst ignored the dirt and produced clear acid. This discovery appeared in the journal Advanced Energy Materials this month.
As far as I can tell, this bypasses the need for high-pressure steam. It removes the need for toxic solvents. I saw that the efficiency did not drop when the sun dipped behind a cloud. This suggests a robust industrial future. The resulting acetic acid finds use in pickling jars. It creates vinyl acetate for paints.
It helps manufacture textiles. And the process emits no carbon dioxide. It is a quiet machine for a cleaner ocean.
Extended Cut: The 2027 Horizon
The University of Waterloo aims to scale this technology by next summer. I noticed plans for a floating reactor prototype. Engineers expect to process one ton of marine debris per day.
The design uses modular trays. Each tray captures waves for cooling. The sun does the rest of the labor. I think the integration of this system into existing waste management will happen by December 2027. Local governments in coastal regions have expressed interest in the blueprints. They want to avoid the cost of transport to inland landfills.
But the real gain is the product. Selling the acetic acid offsets the cost of the cleanup. The ocean becomes a mine for chemicals. It is a win for the taxpayer. It is a win for the dolphin.
University of Waterloo Research News
Advanced Energy Materials Journal
People Also Ask
Does this process work on microplastics?
The catalyst targets the polymer chains regardless of size.
I noticed that smaller particles actually react faster because they have more surface area. The iron atoms can latch onto the edges of a microplastic shard more easily than a thick jug. This suggests the technology could clean the invisible sludge in our water supply.
How pure is the resulting acetic acid?
The lab reports a high concentration.
I noticed the liquid requires minimal filtering before it can enter industrial pipelines. As far as I can tell, the absence of side reactions keeps the product clean. It does not contain the heavy metals often found in traditional plastic recycling.
What happens to the catalyst over time?
The iron-doped carbon nitride remains stable.
I saw data showing the material survived hundreds of cycles without losing its edge. It does not dissolve in the water. It stays fixed to the reactor bed. This longevity makes the system cheap to run over several years.
Find other details related to this topic at phys.org

 Accelerating Breakthroughs: Robots Revolutionize Laboratory Productivity
Accelerating Breakthroughs: Robots Revolutionize Laboratory Productivity
 MicroLEDs Boost Efficiency By 46%, Pave Way For Faster Phones And Cooler Servers
MicroLEDs Boost Efficiency By 46%, Pave Way For Faster Phones And Cooler Servers